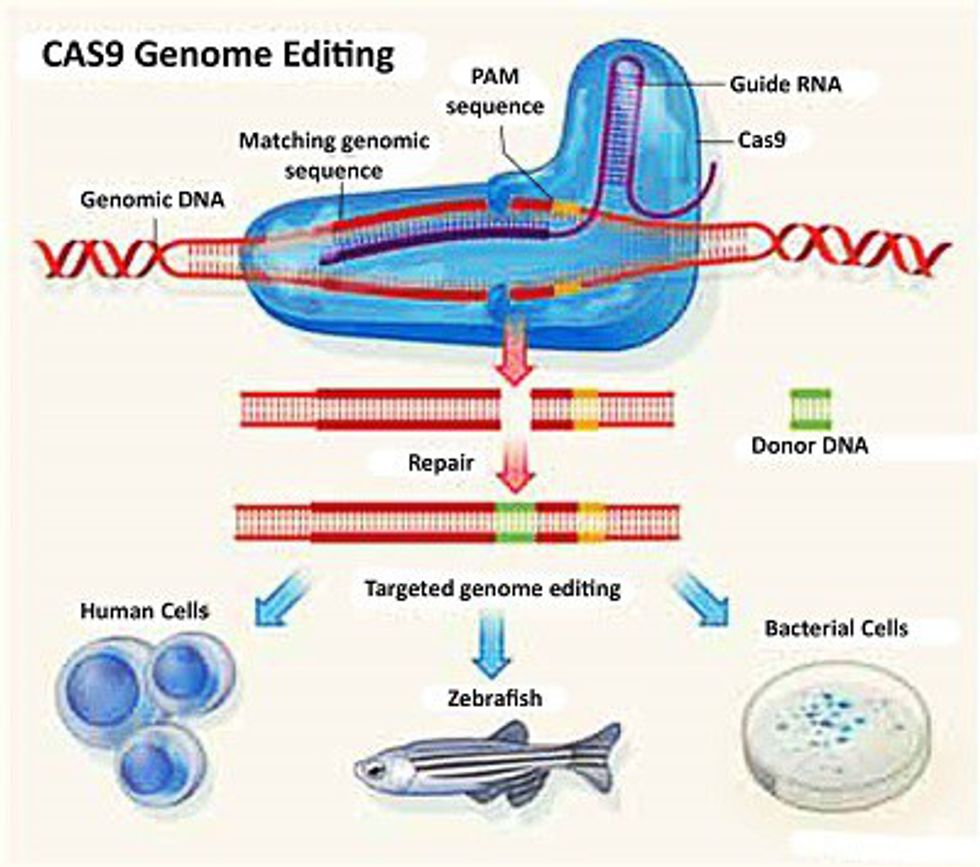
This week, scientists in England applied to begin the process of editing human embryos’ DNA. The application comes on the heels of the controversial use of a new technology called CRISPR-CAs9, developed earlier this year by Chinese scientists who intend to use it to edit human DNA. CRISPIR-Cas9 (known commonly as CRISPR) has been in medical headlines several times since its development last year. The controversial technology has caused a rippling effect in the medical community so large that there will be an international discussion regarding the ethical use for research and implementation of the technology later this year in Washington, D.C.
CRISPR can be thought of as an editing machine in a way. Without the medical jargon, basically CRISPR allows us to remove specific genes from embryos in the germ cells. Why is that important? Because unlike previously, the removal of the gene is actually passed on to the offspring; in previous years we could edit the human genome but it would only affect the person without also passing the edited version of the genome to the offspring. Basically by changing the germ cells' DNA in embryos we are altering the genome with permanent consequences.
Scientists view the CRISPR technology as an intriguing and interesting technology but also approach it with an extremely cautious view. For example, the CRISPR technology has the ability to remove diseases in embryos, so we could very realistically remove the genes that cause sickle cell disease. This sounds great at first, but by removing those genes we could cause a second unintended effect. Like, for example, by removing sickle cells (which are found in higher rates in people who live in Africa where malaria is highly prevalent) we could cause people to become more susceptible to malaria which could potentially lead to more malaria related deaths. Sickle cell is a natural defense to malaria which attacks the blood cells; if the blood cells are deformed, like they are in sickle cells, then malaria has a much harder time attack the blood cells. This is only one example that could be a potential side effect of changing the human genome. Moreover, this change would be passed on to future generations who would lose that defense.
The proponents of CRISPR say that while there are potentially unknown and dangerous side effects, the benefits outweigh them. For example, proponents say that by doing research with DNA editing in embryos we will have a chance to gain an intricate view and understanding of the development of embryos, which would open the doors to a world of possibilities—possibilities that may include a deeper understanding of developmental diseases that can lead to Down syndrome and syndrome X, for example. The proponents want to advocate for use in laboratories on embryos that would not reach development in clinical trials but rather remain in laboratories under regulations. And that is exactly what they will argue for in the international meeting in December in Washington, D.C.
While scientists in China have reported using CRISPR to edit the genome of human embryos, the rest of the world is playing catch-up. Scientists, doctors, and research firms are struggling to grasp the depth of possibility that CRISPR has and will provide and the effects it could have both now and in the future, both intended and unintended. There is no general consensus either, further complicating the issue. The ethics have yet to be decided upon and that is the goal of the meeting in December between the American, British, and Chinese leaders in medical research.
CRISPR can be viewed as a Pandora ’s Box: To some extent, we know that there is potential, but we also don’t understand the consequences that opening the potential could cause. And while the meeting in December may set the parameters for our ethical use of CRISPR, it will fail to put into words whether we can do more good than harm with a technology we only understand in a technical sense. Hindsight, after all, is always 20/20.